
In 1904, Thomson proposed the “plum pudding” model of atoms, which described a positively charged mass with an equal amount of negative charge in the form of electrons embedded in it, since all atoms are electrically neutral. (c) In the cathode ray, the beam (shown in yellow) comes from the. It was given by famous scientist JJ Thomson after the discovery of electrons from his cathode ray experiment. (b) This is an early cathode ray tube, invented in 1897 by Ferdinand Braun. Dalton’s Atomic Theory FAQs on Thomson’s Atomic Model Q1: What is Thomson’s Atomic Model Answer: Thomson’s Atomic Model was the first model to explain the basic structure of the atom. Thomson produced a visible beam in a cathode ray tube. Thomson in 1904, following his discovery of the electron in 1897 after his famous cathode ray experiment.
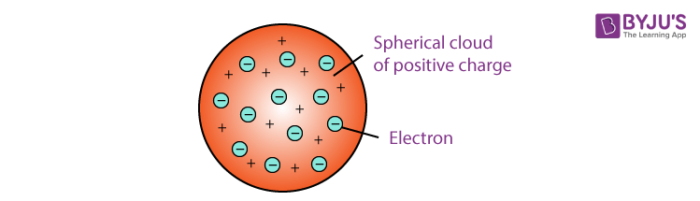
However, the positively charged part of an atom was not yet well understood. The results of these measurements indicated that these particles were much lighter than atoms (Figure 2.2.1 2.2. Thomson’s Atomic Model was proposed by J.J. He enrolled at Owens College, Manchester, in 1870, and in 1876 entered Trinity College, Cambridge as a minor scholar. Scientists had now established that the atom was not indivisible as Dalton had believed, and due to the work of Thomson, Millikan, and others, the charge and mass of the negative, subatomic particles-the electrons-were known. Joseph John Thomson was born in Cheetham Hill, a suburb of Manchester on December 18, 1856.
